Islet vascularized ECM gel (IVEG) construct for islet culture and transplantation
Daniel M Tremmel1, Vansh S Jain1, Samantha A Mitchell1, Sara D Sackett1, Jon S Odorico1.
1Surgery - Transplantation, University of Wisconsin, Madison, WI, United States
Introduction: Islet isolation results in destruction of the extracellular matrix (ECM), also severing connections with endothelial cells (ECs). Following transplantation, islets re-vascularize, enabling survival and function in vivo, but the time between isolation and revascularization results in significant islet loss. Furthermore, much that is known about islet biology is based on research using isolated islets, which are missing crucial connections and signals from the native environment. We developed a culture system that reconstructs the islet ECM and creates a network of capillary-like endothelial tubes in vitro, prior to transplantation. The islet vascularized ECM gel (IVEG) enables the study of crosstalk between islets and the islet microenvironment in vitro. IVEG constructs can also be easily handled for transplantation studies.
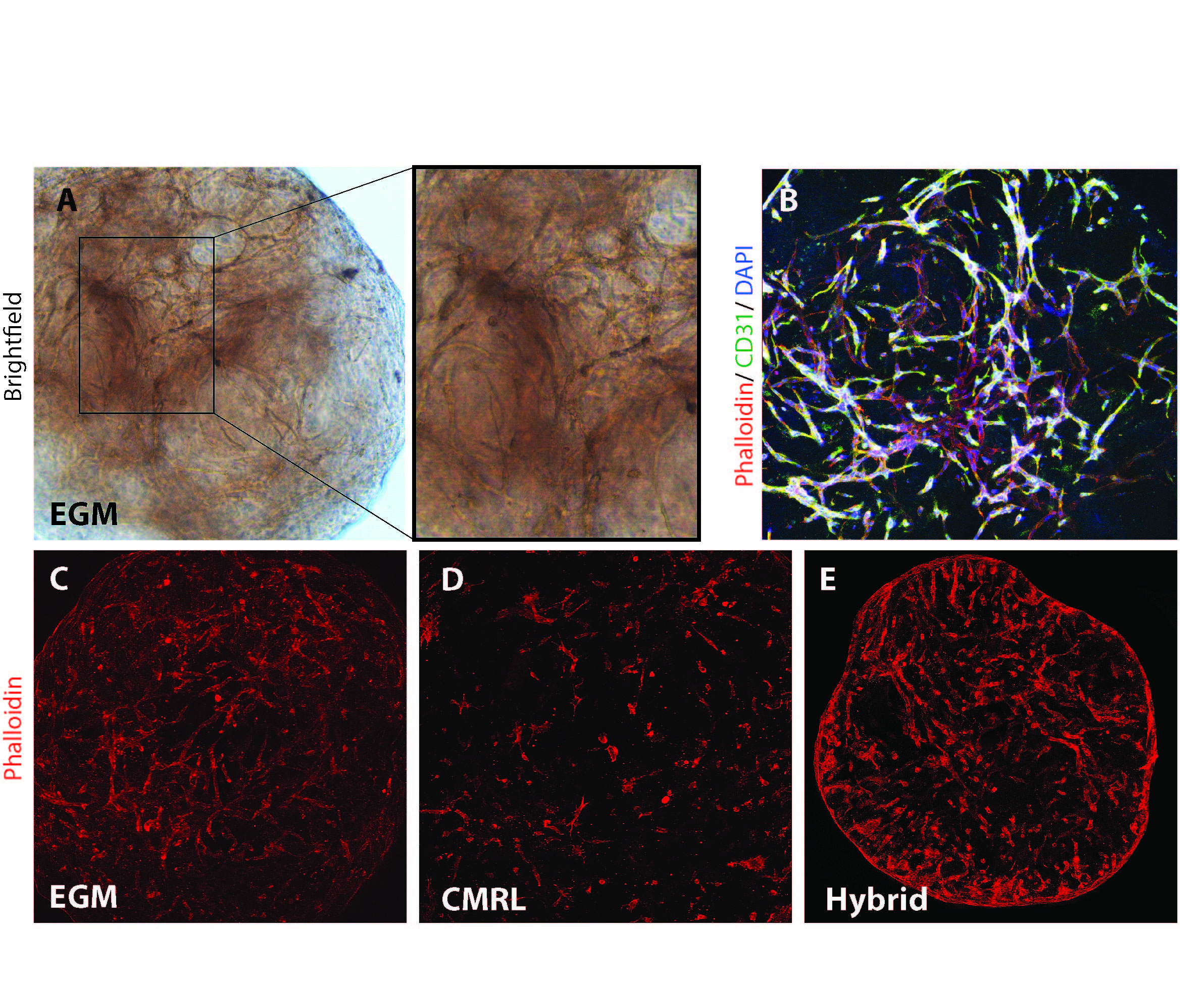
Methods: Human pancreatic ECM hydrogel (hP-HG) was prepared by decellularization of pancreas, solubilization of the ECM, and neutralization to form a gel. Human ECs (HUVECs) were cultured in various islet, EC, and hybrid media to optimize 3-D tube formation (Fig 1A-B). Human islet function was assessed in IVEG constructs (Fig 2A-B) under ideal media conditions after 3 days of culture through static GSIS.
In a pilot study, human islets were transplanted subcutaneously (SQ) in diabetic immunodeficient mice (NSG RIP-DTR) either alone, or within IVEG constructs to assess graft function.
Results: ECs cultured in hP-HG in EC growth medium (EGM) formed 3D tubes covering 29% of the area of a max-intensity image (Fig 1C). When cultured in islet medium (CMRL), the percentage was reduced (14.2%) (Fig 1D), but when cultured in a hybrid medium combining EC and islet media, the vessel coverage was 31.7% (Fig 1E). The combination of these media and methods enables the co-culture of endothelial tubes with islets in hP-HG for several days.
Islets cultured in hP-HG alone had a significantly improved stimulation index (SI = 7.6) compared to the same donor islets cultured in suspension (SI = 2.1). Islets cultured in medium with EC growth factors with or without added ECs had a stimulation index comparable to that of islets in hP-HG alone (SI = 10.0). (Fig 2C) The gel constructs can easily be transplanted into the kidney capsule (Fig 2D) or subcutaneously (Fig 2E).

Diabetic mice did not experience stable serum insulin levels following SQ transplantation with 1000 IEQ alone, and did not have reductions in fasting or fed blood glucose (BG) levels, while mice transplanted with the same donor islets after 3 days of IVEG culture had reductions in both fasting and fed BG levels, and stable human insulin detected in serum (N=3 mice per group).
Conclusions: ECs are able to form 3-D tubes in hP-HG with maintained islet viability and function after 3 days of co-culture. This culture system enables the study of islet-endothelial cell interactions in vitro. Future studies will test how the IVEG constructs engraft and function after transplantation.